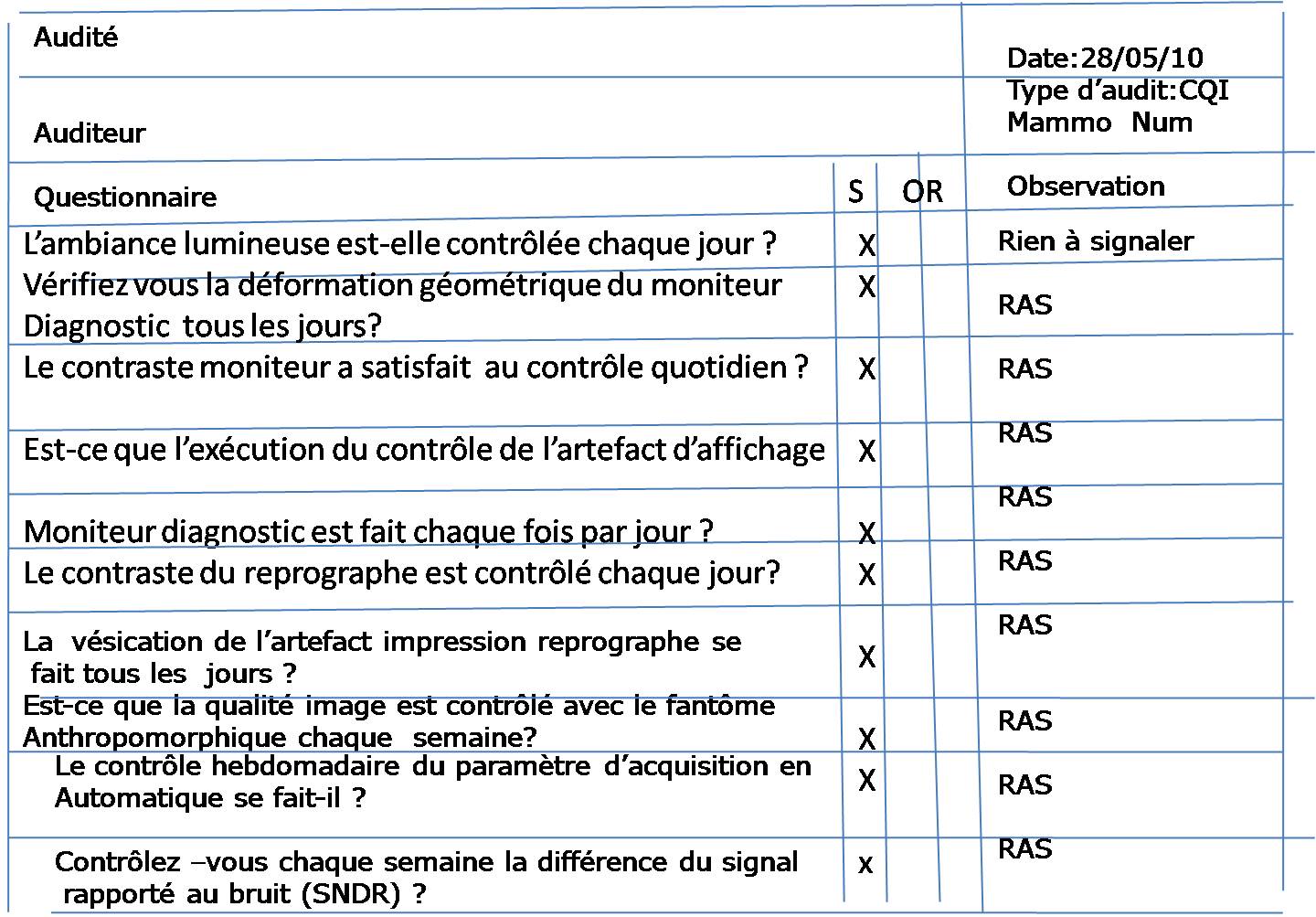
GMP is normally applied in food, beverages, pharmaceuticals, dietary supplements, cosmetics, and medical devices. It aims to minimize the risks of health and safety concerns involved in any production that cannot be eliminated through production and final testing of the product. Furthermore, it conforms to guidelines that control the authorization and licensing of the manufacture and sale of products. Good Manufacturing Practices is a system designed to guarantee that the production of goods adheres to a country’s set of standards. A Crash Course on Good Manufacturing Practices As a solution, QSE Academy compiled all the information you need regarding GMP auditing and the tasks of an auditor. 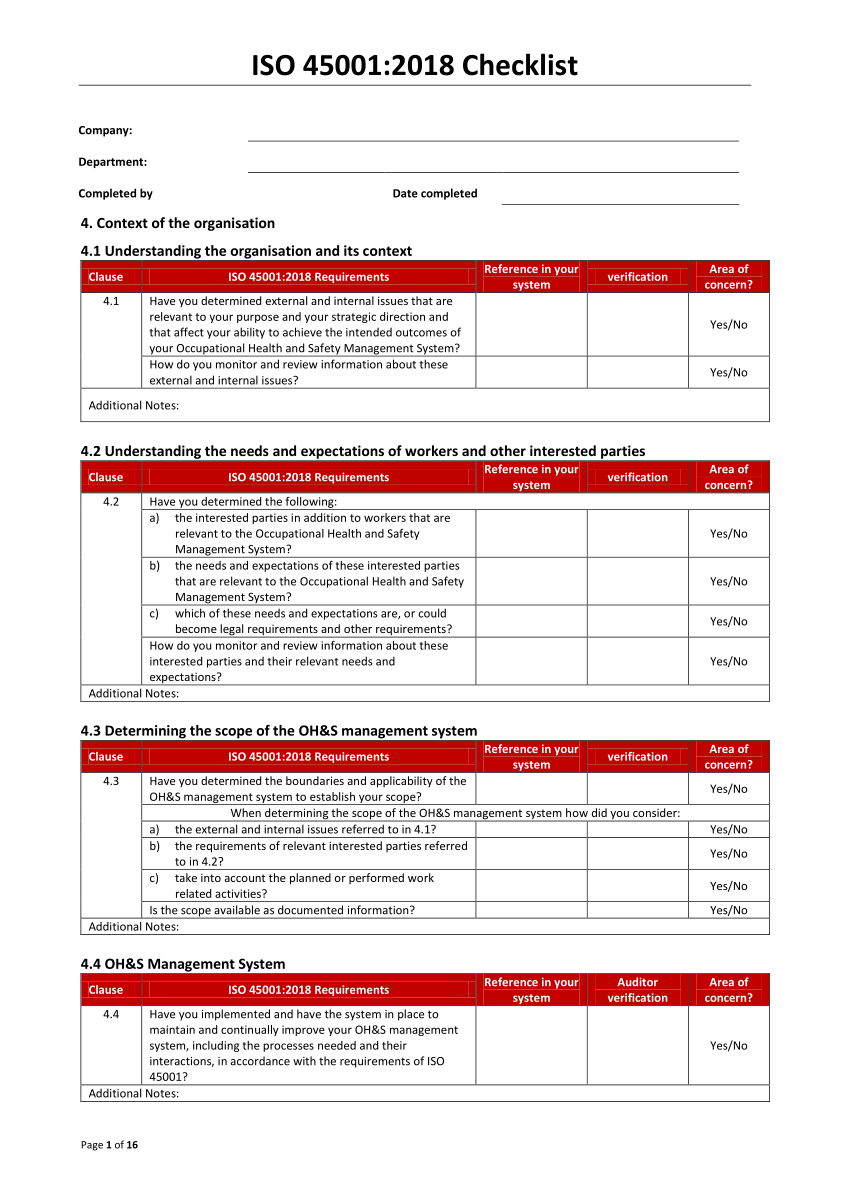
The GMP audit is meticulous and requires a capable auditor. Good Manufacturing Practices is a scheme for ensuring products are produced according to quality standards and conform to guidelines.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
September 2023
Categories |
 RSS Feed
RSS Feed
